Wood Pallet Recycling for Profit: http://tinyurl.com/23o36cr
Great product, $32.50/sale. Package of five .pdf files, 2 .xls and 6 .mp4 videos. Great offline business for anyone with a truck, or even a station wagon. Pallets are everywhere, for free, and can be sold for lots of money.
Antique Furniture - How to Find It and Fix It!: http://tinyurl.com/3y2lpfa
Antique Furniture can bring history and interest into your home, or money into your wallet, if you know what to look for, how to find it and what to do with it once it's yours! This 100 page guide shows you all this and more. 3 Fantastic Bonuses - Free!
My site for all the good gear I find about precious metal refining and anything else to do with scrap metal. If you think of something to contribute, please comment or contact me.
Friday, November 12, 2010
Sunday, November 7, 2010
Saturday, November 6, 2010
Interesting Info about Nitric Acid
The red gas which was formed is Nitrogen Dioxide, sometimes other gases form at the same time, so we just call it NOx. Copper nitrate has a lot of uses, especially in explosives. That is why Nitric Acid is a very strictly controlled acid.
Saturday, October 23, 2010
Why Are You Keeping All the Clutter?
You know what the real problem is with getting organized?
We have too much stuff!
I\'m willing to bet you have some things you (or someone you live with) keep without having a good enough reason.
So why DO we keep so much stuff?
The thing is, it\'s easier to find excuses for why you should keep something and delay making a decision rather than making a firm (and sometimes difficult) choice to say goodbye to your stuff.
Here are four of my favorite excuses...
1. I might need it someday.
Yes, you might. But the thing is most of the items we keep can easily be found or replaced within a day or two. Lots of men (my dad included) keep every screw and nail created and store it away like a squirrel with his nuts.
But what happens is these little tiny items create more and more clutter and it gets to a point where you can\'t find that little screw anyway because it\'s like finding a needle in a haystack.
2. I\'m going to lose some weight and start wearing this again.
I hope if you have a goal to lose some weight, you do everything in your power to make it happen. And when you do, I give you permission to go out and find a sale and buy some brand new clothes. You should be proud and it\'s the perfect time to reward yourself.
3. So-and-So Gave Me This.
I\'m all for keeping memories and items that remind us of people we love.
But the truth is, memories are not in the clutter, the knick-knacks and stuff you have shoved in a box.
I won\'t say get rid of everything and it\'s not always an easy decision. But try and just keep the special things you cherish.
And get them out in the open, on display - where you can enjoy them and have a story to tell when someone comments or asks a question about the item.
4. I paid good money for this... thing!
I\'m sure you did. But the thing is, what has more value...this item you no longer have any use for or the way you enjoy your house? The item...or your happiness? The item... or your space?
5. Oh, I\'m just trying to figure out what to do with it...
Okay, so maybe it\'s not worth holding onto.
See, if it takes that much mental work to figure out what to do with something, you can take a pretty good guess that it may not be worth as much to you as you think.
If you can\'t figure out what to do with something...it\'s a safe bet you won\'t miss it too much.
There is a lot more value in enjoying your home, your friends and your family than the stuff you paid for.
How do I know what type of gold, platinum or silver I have?
Pure gold is 24carat and many countries use pure gold, or 22ct for their jewellery, but generally speaking the Australian market prefers the look of gold mixed with alloys.
This process is quite regulated and jewellery should have a stamp mark indicating the gold content.
For example:
9ct gold is stamped either9ct, 9k, 375.
10ct gold is stamped either 10ct, 10k, 416.
14ct gold is stamped either14ct, 14k, 585.
18ct gold is stamped either18ct, 18k, 750
21/22/23/24 Carat gold quite often will not have a stamp mark
Platinum – stamped 950, 950PT, Plat
Silver – 925, StgSil, SSil, Stg. (Most silver jewellery is Sterling Silver)
Other silver – 800.
Jewellery marked with GF, GP, or RG are gold plated
Jewellery marked with 9ct/StgSil, 9k/925 or variations mean that you have a 9ct gold sleeve with silver inside; this can apply to chains, bracelets and bangles.
I suggest you visit www.goldtester.net and www.HowToTestGold.com for more information on testing Gold.
and this RSS feed, which I will add to my website gives the latest precious metal scrap prices.
Friday, August 27, 2010
Interesting videos from nurdrage.com on Nitrc acid Production
Here are a couple of Handy videos worth watching regarding testing nitrate fertilizer and making nitric acid. All credit for them goes to nurdrage.com and their website has more interesting info.
In this video we show you how to test if a fertilizer has nitrates rather than urea or ammonia as its nitrogen source.
The idea is quit simple, in a strongly acidic solution the nitrates will behave like nitric acid and dissolve copper metal releasing nitrogen dioxide gas. Looking for this gas is a strong indication for the presence of nitrates.
WARNING: these reactions produce toxic nitrogen dioxide gas, this must be performed outside or in a fume hood.
To perform the test, simply mix some of your fertilizer with water to dissolve the nitrates and then mix it with hydrochloric acid. Then add in some copper metal.
The reaction is very slow to start up unless you heat it. A nerdy way of heating it on the spot without using a heater is to add a small ball of aluminum metal, don't use too much or it will go out of control. Stir the mixture as the aluminum dissolves to distribute the heat evenly.
After the mixture is heated, place a glass container over the mixture to keep the gases in. If nitrogen dioxide is forming the distinctive brown nitrogen dioxide will fill the container.
And that's how you can tell if there are nitrates in your fertilizer.
If you want to test for ammonia (in case you have ammonium nitrate) just mix some fertilizer with half as much sodium hydroxide and add a little water to cover it. If it starts bubbling and releasing ammonia gas then it contains ammonia.
3 ways to make nitric acid based on two different chemical approaches both of which can be done using easily accessible materials.
Warning: The procedures in this video produce large quantities of toxic gases and deal with highly corrosive acids. All work must be performed in a fume hood with proper safety equipment. And all apparatus must be glass to withstand the acids.
Chemically, nitric acid is made by bubbling nitrogen dioxide into water. So the objective in this approach is to generate nitrogen dioxide. This can be done by reacting hydrochloric acid, a nitrate salt and copper. Around 80grams of sodium nitrate, over 30 grams of copper and 100mL of hydrochloric acid (37% 12M) are the quantities needed. The exact amount isn't critical. For usable concentrations, the amount of water being converted should be small, around 20-50mL.
Any source of nitrate is usable including potassium nitrate, ammonium nitrate and even nitrate-based fertilizers. You can use our previous video on testing for nitrates if you want to determine if yours can be used. http://www.youtube.com/watch?v=f5M3rU...
The tricky part now is leading the gas into water. Two approaches are shown in the video. In the first approach three containers, such as jars are place inside each other to force the gas to go into the water. This is very inefficient but is very simple to do.
The better approach is to lead the gas out of the generator through a tube and into a chilled container of water.
The water that's converted into nitric acid can be replaced with hydrogen peroxide for better yield.
The chemical waste that's generated contains the valuable copper used before and recovering it is worthwhile due to todays high copper prices. This is simply done by putting in enough aluminum metal that it reacts with all the acids and copper in solution to create a slurry of copper. This can be filtered to obtain a residue of copper. Its highly contaminated but can still be used to make more nitric acid.
For further information on the chemistry type "copper and nitric acid" into google. The hydrochloric acid and nitrate salt behave as nitric acid (with nitrate from the salt and protons from the hydrochloric acid) and dissolve the copper releasing nitrogen dioxide gas.
You can use other concentrations of hydrochloric acid but you need to decrease the amount of water added to keep the concentrations the same.
Finally, the last way of making pure nitric acid is to react concentrated sulfuric acid and a pure nitrate salt (NOT fertilizer) and heat it in a glass distillation apparatus to distill over the pure nitric acid. Stoichiometric quantities of both reagents are recommended for maximum yield.
Friday, August 20, 2010
Magnets - AMF magnetics
A good magnet is essential for scrap metal work to sort the ferrous from the non-ferrous. Check out the huge range in the AMF catalog for some ideas.
Sunday, July 25, 2010
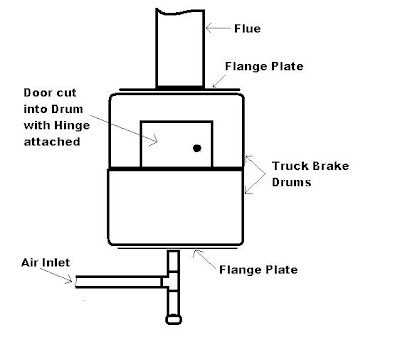
The Brake Drum Forge
My Father has been very successful in smelting metals using a simple wood fired forge made from 2 discarded truck brake drums and some other scrap metal bits and pieces.
The Basic construction for this type of forge is as per the rough diagram below.
The Door in the top Drum was cur using an Angle grinder, then the hinge and drums where welded together using low carbon welding rods. (Normal Mild Steel welding rods do not work well on this kind of metal).
The Door could be cut with oxy/acetalene but this is very messy and difficult so I would not recommend it. The Air source we used was an old hairdryer, and we have used a vacuum cleaner as well, both easy to get and cheap.
A small steel crucible was used to melt aluminum and lead with ease, and this furnace could easily smelt iron or steel, but a proper silicon carbide type crucible would be required.
As Truck brake drums are designed to withstand heat, they tend to last quite a few years before burning out (we haven't burnt one out yet).
I'd be curious to see what other people have used to make their own furnaces!.
Food for thought.
Friday, July 16, 2010
Hows the weather
More interesting info in the works, please forgive my recent non posting spree. I'll make it up to you I promise :-)
Saturday, July 10, 2010
Bits - for the weekend. More good recycling stuff coming.
Here are a few thoughts for the weekend...
Wealthy people miss one of life's greatest thrills . . . Making the last car
payment.
Always arrive late at the office, but make up for it by leaving early.
You can get more of what you want with a kind word and a gun than you can with just a kind word.
Buffet; A French word that means "get up and get it yourself.
Airplane travel is nature's way of making you look like your passport photo.
When all else fails. Follow instructions.
Those are my principles. If you don't like them I have others.
Good judgment comes from bad experience, and a lot of that comes from bad judgment.
Copy from one, it's plagiarism; copy from many, it's research.
I love deadlines. I especially love the whooshing sound they make as they fly by.
If ignorance is bliss, why aren't there more happy teenagers?
No one will win the battle of the sexes, there is too much flirting with the
enemy.
Some people ask the secret of our long marriage. We take time to go to a
restaurant two times a week. A little candlelight, dinner, soft music and
dancing. She goes Tuesdays, I go Fridays.
As you get older three things happen. The first is your memory goes, and I can't remember the other two...
Looking For Speaking Success?: http://tinyurl.com/29vscjn
Parenting Book - How To Parent Together!: http://tinyurl.com/26ow9or
Tarot Card Reading Secrets.: http://tinyurl.com/2eqp2cb
Parenting Book - How To Parent Together!: http://tinyurl.com/26ow9or
Tarot Card Reading Secrets.: http://tinyurl.com/2eqp2cb
Friday, July 2, 2010
Did you Know?
SILVER
Over 95% of the silver produced in a year is used in photography, jewelry and silverware, and industrial products
Kodak and Fuji companies are the world's largest consumers of silver
Silver is effective in treating burns by killing bacteria, allowing the burn to heal more quickly pure silver has the highest electrical and thermal conductivity of all metal.
The main producers of silver are the United States, Mexico and South America
GOLD
Gold was made into art objects and jewelry as far back as 4000 BC
Alchemists, the forerunners of today's chemists, attempted to turn base metal into gold
Gold does not tarnish, rust or corrode
One cubic foot of gold weighs more than half a ton
The main producers of gold are South Africa, the United States, Australia, China, Canada, and Russia
PLATINUM
Platinum is the rarest of all precious metals
Approximately 10 tons of raw ore have to be mined to produce one pure ounce of platinum
The main producers of platinum are the former Soviet Republics, South Africa, and Canada
All the platinum ever mined would fill a cube less than 25 feet on each side
Scarce metals, such as platinum, face depletion risks this century because of the lack of suitable substances in such devices as catalytic converters and hydrogen fuel cells.
Sunday, June 6, 2010
Reverse Electroplating Gold and Platinum.
I will be adding a lot more in this interesting topic into my scrap metal adventure site,
heres some snippets to feed the mind...
The anodes for gold and platinum plating are typically platinized titanium, so that material can be used as the cathode in the setup you are describing -- but stainless steel is usually good enough, depending on the particular plating bath.
For electrodeposition of gold and other metals you need a high Current , low voltage power source..
2.5 V, 20 A. CIRCUIT FOR ELECTROPLATING
My thoughts would be to use the power supply from an old pool clorinator as it will be similarly high current, low voltage.
This type of electroplating technique would need to be performed in a ceramic or fiberglass container devoid of metals.
" The simplicity refiner from Shor to refine karat gold. The alloy is dissolved into a saltwater solution by using either a rectifier or battery charger (12 v 10 amp+) then the gold is precipitated by adding a powdered chemical (I believe is sodium metabisulfite). The "mud" is collected rinsed and dried and melted back to 999.5+ purity. "
For electrodeposition of gold and other metals you need a high Current , low voltage power source..
2.5 V, 20 A. CIRCUIT FOR ELECTROPLATING
My thoughts would be to use the power supply from an old pool clorinator as it will be similarly high current, low voltage.
This type of electroplating technique would need to be performed in a ceramic or fiberglass container devoid of metals.
" The simplicity refiner from Shor to refine karat gold. The alloy is dissolved into a saltwater solution by using either a rectifier or battery charger (12 v 10 amp+) then the gold is precipitated by adding a powdered chemical (I believe is sodium metabisulfite). The "mud" is collected rinsed and dried and melted back to 999.5+ purity. "
Saturday, May 29, 2010
PC gold recovery, a profitable business!
Scrapped computer components are fine sources of scrap materials, especially scrap gold. PC gold recovery has become a profitable business.
Actually computer processors are good gold sources, while other scrapped components from your old numbers cruncher produce their own valuable metals and other recyclable materials. Memory sticks with their gold tip scrap are taking an increasing part of the computer scrap market. In sort: there's quite a lot of (scrap) gold in computers.
Wednesday, May 5, 2010
My main site updated
I've finished migrating Metal Scrap Adventure - all well with the world again. Click here to check it out.
now time for a joke: Aussie style :-)
A bloke's wife goes missing while diving off the West Australian coast. He reports the event, searches fruitlessly and spends a terrible night wondering what could have happened to her.
Next morning there's a knock at the door and he is confronted by a couple of policemen, the old Sarge and a younger Constable. The Sarge says, 'Mate, we have some news for you, unfortunately some really bad news, but, some good news, and maybe some more good news'.
'Well,' says the bloke, 'I guess I'd better have the bad news first?' The Sarge says, 'I'm really sorry mate, but your wife is dead. Young Bill here found her lying at about five fathoms in a little cleft in the reef. He got a line around her and we pulled her up, but she was dead.'
The bloke is naturally pretty distressed to hear of this and has a bit of a turn. But after a few minutes he pulls himself together and asks what the good news is. The Sarge says, 'Well when we got your wife up there were quite a few really good sized crays and a swag of nice crabs attached to her, so we've brought you your share.' He hands the bloke a sugar bag with a couple of nice crays and four or five crabs in it.
'Geez thanks. They're bloody beauties. I guess it's an ill wind and all that... So what's the other possible good news? 'Well', the Sarge says, 'if you fancy a quick trip, me and young Bill here get off duty at around 11 o'clock and we're gonna shoot over there and pull her up again!
now time for a joke: Aussie style :-)
A bloke's wife goes missing while diving off the West Australian coast. He reports the event, searches fruitlessly and spends a terrible night wondering what could have happened to her.
Next morning there's a knock at the door and he is confronted by a couple of policemen, the old Sarge and a younger Constable. The Sarge says, 'Mate, we have some news for you, unfortunately some really bad news, but, some good news, and maybe some more good news'.
'Well,' says the bloke, 'I guess I'd better have the bad news first?' The Sarge says, 'I'm really sorry mate, but your wife is dead. Young Bill here found her lying at about five fathoms in a little cleft in the reef. He got a line around her and we pulled her up, but she was dead.'
The bloke is naturally pretty distressed to hear of this and has a bit of a turn. But after a few minutes he pulls himself together and asks what the good news is. The Sarge says, 'Well when we got your wife up there were quite a few really good sized crays and a swag of nice crabs attached to her, so we've brought you your share.' He hands the bloke a sugar bag with a couple of nice crays and four or five crabs in it.
'Geez thanks. They're bloody beauties. I guess it's an ill wind and all that... So what's the other possible good news? 'Well', the Sarge says, 'if you fancy a quick trip, me and young Bill here get off duty at around 11 o'clock and we're gonna shoot over there and pull her up again!
Thursday, April 22, 2010
I'm Curious
I've managed to separate a small amount of gold flake and am going to boil off the water and flame it. I wonder if Gold Buyers Australia can test this for purity? I must ask them:-)
Sunday, April 11, 2010
Gold Refining from scrap
There are several methods I’ve found to separate precious metals from other metals.
To dissolve Copper from gold plated printed circuits or plated Brass pins / connectors.
CAUTION: Remember the golden rule, acid to water, not water to acid to prevent splash injury.
If plated heavy I use a homemade nitric acid--- sodium nitrate (Na(NO3)2) and battery acid (H2SO4) 1:1 ratio, I use store bought H2SO4 as it contains no lead like that recovered from car batteries, this gives HNO3 + Na SO4 +h2O
Straight Hydrochloric Acid (HCL) also works at dissolving Copper but much slower.
Also I’ve read you can use Hydrochloric Acid mixed 1:1 with Hydrogen Peroxide. This is a chemical used by some hobbyists to etch printed circuit boards but the reaction when the 2 chemicals are mixed causes a lot of heat so plastic containers CANNOT be used, Glass or crock-pot is best.
Ferric Chloride (Hydrochloric Acid mixed with Rust) and Ammonia will also dissolve copper as these are also used for etching circuit boards.
If scrap gold is mixed with other metals separate as much as possible by cutting open parts to expose base metals
Small pieces work best
The next step should be performed outside on hot plate or in a crock-pot (not a metal or plastic container). Put acid then gold then sodium nitrate (can also use ammonium nitrate fertilizer or potassium nitrate sold as stump remover)
Boil this
CAUTION: stay clear of fumes and if possible a fume hood is advisable. Not inside. Also very corrosive to metals.
This will make a nitric acid that will dissolve copper and other metals leaving gold in a salt metal blue / green solution of copper sulfate and gold flakes,
Add water till all salt in liquid, and let the gold flakes settle.
Take a syringe and remove remaining liquid, save for copper electrolysis or add lesser active metal like Zink or aluminium to remove copper and then baking soda or caustic soda to neutralize the acid.
Copper can also be precipitated out of solution using Sodium Hydroxide (caustic Soda).
I found out that the casings of old computer chips are made with an aluminium based compound which I assume is for heat exchange reasons. I put some in the solution containing copper and it precipitated the copper straight out and the copper settled to the bottom.
These types of chips require Aqua Regis to strip the gold.
If there was Silver in the metal, NaCL or table salt will make it precipitate out as white cottage cheese powder, you can take these oxides and add a little bit of borax soap as flux and melt back to metal with a gas torch.
For More Complex Gold Plated items like computer processors.
Use Home made Aqua Regis to dissolve gold by using the above nitric Acid and hydrochloric acid (muriatic used to clean concrete or in swimming pools), 2 parts Nitric Acid (HNO3 + Na SO4 +h2O) to 1 part Hydrochloric acid (HCL)
CAUTION: Aqua Regis (AR Agent) is extremely corrosive and will dissolve all metals with the exception of some rare earth metals like titanium.
Boil this to speed up removal of nitric acid as red cloud in the liquid.
CAUTION: stay clear of fumes and if possible a fume hood is advisable. Not inside.
Boil until almost but not dry adding a little Hydrochloric acid (HCL) to wet. Do this three times then add 10 times the amount of distilled water.
Silver if any should precipitate as white powder, as per previous a little table salt will make the silver precipitate.
Then make a solution of ferrous (Iron) sulphate from old cut up transformer iron or nails/steel wool, sulphuric acid and seven times water. Wait till this solution turns a greenish colour.
Ferrous Sulphate added to aqua Regis gold solution will make gold fall to bottom as a brown powder let settle at least 24hrs.
Remove liquid solution of copper or other metals and just like the previous example save for copper electrolysis or add lesser active metal like Zink or aluminium to remove copper and then baking soda or caustic soda to neutralize the acid.
Take the brown dried powder, add a little borax and torch to turn back to gold.
The chemicals like Aqua Regis if purchased will usually cost more than the gold is worth.
Aqua Regis will also dissolve Platinum, but I have yet to try this. I would be interested in finding if this can be used to extract the Platinum from Catalytic Converter materials…
I SAY AGAIN CAUTION: THIS IS DANGEROUS CHEMISTRY, AND ALL PRECAUTIONS MUST BE TAKEN IF ATTEMPTING THIS KIND OF ACTIVITY.
To dissolve Copper from gold plated printed circuits or plated Brass pins / connectors.
CAUTION: Remember the golden rule, acid to water, not water to acid to prevent splash injury.
If plated heavy I use a homemade nitric acid--- sodium nitrate (Na(NO3)2) and battery acid (H2SO4) 1:1 ratio, I use store bought H2SO4 as it contains no lead like that recovered from car batteries, this gives HNO3 + Na SO4 +h2O
Straight Hydrochloric Acid (HCL) also works at dissolving Copper but much slower.
Also I’ve read you can use Hydrochloric Acid mixed 1:1 with Hydrogen Peroxide. This is a chemical used by some hobbyists to etch printed circuit boards but the reaction when the 2 chemicals are mixed causes a lot of heat so plastic containers CANNOT be used, Glass or crock-pot is best.
Ferric Chloride (Hydrochloric Acid mixed with Rust) and Ammonia will also dissolve copper as these are also used for etching circuit boards.
If scrap gold is mixed with other metals separate as much as possible by cutting open parts to expose base metals
Small pieces work best
The next step should be performed outside on hot plate or in a crock-pot (not a metal or plastic container). Put acid then gold then sodium nitrate (can also use ammonium nitrate fertilizer or potassium nitrate sold as stump remover)
Boil this
CAUTION: stay clear of fumes and if possible a fume hood is advisable. Not inside. Also very corrosive to metals.
This will make a nitric acid that will dissolve copper and other metals leaving gold in a salt metal blue / green solution of copper sulfate and gold flakes,
Add water till all salt in liquid, and let the gold flakes settle.
Take a syringe and remove remaining liquid, save for copper electrolysis or add lesser active metal like Zink or aluminium to remove copper and then baking soda or caustic soda to neutralize the acid.
Copper can also be precipitated out of solution using Sodium Hydroxide (caustic Soda).
I found out that the casings of old computer chips are made with an aluminium based compound which I assume is for heat exchange reasons. I put some in the solution containing copper and it precipitated the copper straight out and the copper settled to the bottom.
These types of chips require Aqua Regis to strip the gold.
If there was Silver in the metal, NaCL or table salt will make it precipitate out as white cottage cheese powder, you can take these oxides and add a little bit of borax soap as flux and melt back to metal with a gas torch.
For More Complex Gold Plated items like computer processors.
Use Home made Aqua Regis to dissolve gold by using the above nitric Acid and hydrochloric acid (muriatic used to clean concrete or in swimming pools), 2 parts Nitric Acid (HNO3 + Na SO4 +h2O) to 1 part Hydrochloric acid (HCL)
CAUTION: Aqua Regis (AR Agent) is extremely corrosive and will dissolve all metals with the exception of some rare earth metals like titanium.
Boil this to speed up removal of nitric acid as red cloud in the liquid.
CAUTION: stay clear of fumes and if possible a fume hood is advisable. Not inside.
Boil until almost but not dry adding a little Hydrochloric acid (HCL) to wet. Do this three times then add 10 times the amount of distilled water.
Silver if any should precipitate as white powder, as per previous a little table salt will make the silver precipitate.
Then make a solution of ferrous (Iron) sulphate from old cut up transformer iron or nails/steel wool, sulphuric acid and seven times water. Wait till this solution turns a greenish colour.
Ferrous Sulphate added to aqua Regis gold solution will make gold fall to bottom as a brown powder let settle at least 24hrs.
Remove liquid solution of copper or other metals and just like the previous example save for copper electrolysis or add lesser active metal like Zink or aluminium to remove copper and then baking soda or caustic soda to neutralize the acid.
Take the brown dried powder, add a little borax and torch to turn back to gold.
The chemicals like Aqua Regis if purchased will usually cost more than the gold is worth.
Aqua Regis will also dissolve Platinum, but I have yet to try this. I would be interested in finding if this can be used to extract the Platinum from Catalytic Converter materials…
I SAY AGAIN CAUTION: THIS IS DANGEROUS CHEMISTRY, AND ALL PRECAUTIONS MUST BE TAKEN IF ATTEMPTING THIS KIND OF ACTIVITY.
Subscribe to:
Posts (Atom)